BrainGate2 will use brain implants and machine learning to ‘decode’ intended speech of people who can’t speak
(SACRAMENTO)
UC Davis Health is recruiting people for a clinical trial with the goal of building a “neurological prosthesis” for restoring speech to people who have lost — or are losing — the ability to speak. The research is aimed at people who have neurological injuries or progressive neurological diseases, such as spinal cord injury, stroke and amyotrophic lateral sclerosis (ALS).
With this study, UC Davis joins BrainGate, an ambitious consortium of universities and academic medical centers studying how brain-computer interfaces can be used to restore neurological function in people living with paralysis. The technology is also sometimes referred to as brain-machine interfaces or neuroprostheses.
The focus of BrainGate is to use this technology to “turn thoughts into action” for people living with paralysis. Some of the consortium’s studies have turned “neural” handwriting into text output to a computer and helped participants with paralysis control a robotic arm and hand.

David Brandman and Sergey Stavisky are leading the BrainGate2 clinical trial at UC Davis Health.
The researchers at UC Davis are focused on a condition known as anarthria, where people want to speak but are unable to control their vocal cords or mouth in a way that would produce audible speech.
“Losing the ability to speak is devastating. The existing assistive communication technologies available to people living with paralysis, such as eye trackers, and sip and puff devices, are slow, cumbersome, and require substantial effort from both the user and their caretaker,” said David Brandman.
Brandman is an assistant professor in the Department of Neurological Surgery and the site-responsible investigator for the study. He is also the co-director of the UC Davis Neuroprosthetics Lab and will lead the surgeries for the study. “It’s my hope that brain-computer interface technology will one day restore functional independence for people living with paralysis."
Brain areas for speech are intact, but the last output step is lost
With some diseases and injuries, the areas of the brain responsible for language and wanting to speak are intact, but the signals cannot get through to the nerves and muscles that need to receive command impulses to produce sound.
With this new study, the researchers hope to “read” the brain signals meant to move the muscles involved in speaking — the tongue, jaw, lips, voice box and diaphragm — and translate the person’s intention to speak into comprehensible speech produced by a computer.
The researchers explain that the technology cannot read minds or detect inner monologue and thoughts. The investigational technology is being developed only to communicate the signals generated in the brain as part of intentional speech.
“By implanting electrodes that can record from individual brain cells involved in generating speech, we hope we can enable the participants to communicate by just trying to speak,” said Sergey Stavisky.
Stavisky, a neuroscientist and neuroengineer, is an assistant professor in the Department of Neurological Surgery. He is the scientific lead for the study and the co-director with Brandman of the UC Davis Neuroprosthetics Lab.
“There are many open questions. We hope to learn what brain cells do when a person tries to speak. For example, what kind of information does this brain activity contain? How does that relate to the movements or sounds the person is trying to produce? With this study, we anticipate we will be able to answer those and many other questions,” Stavisky said.
Implants are not new, but technology is evolving
Implants have been used in the brain to help with disease symptoms for decades. Hundreds of thousands of people globally have been implanted with deep brain stimulation devices. The technology is routinely applied to help people with Parkinson’s disease, tremor and epilepsy. In deep brain stimulation, implants that generate electric impulses are used to manage symptoms of neurological diseases.
What is different about the BrainGate2 clinical trial is that the implants are not there to stimulate the nerve cells. Instead, they are there to “listen” to them.
Participants accepted into the study will have tiny, specialized devices known as Utah Arrays implanted in the region of their brain that generates speech. The U.S. Food and Drug Administration has provided permission for testing the devices in clinical trials.
The arrays obtain impulses from the nerve cells — neural recordings — that are conveyed to two small metal pedestals on top of the head. The pedestals are about a half-inch in diameter. From there, the signals can be connected to a computer.
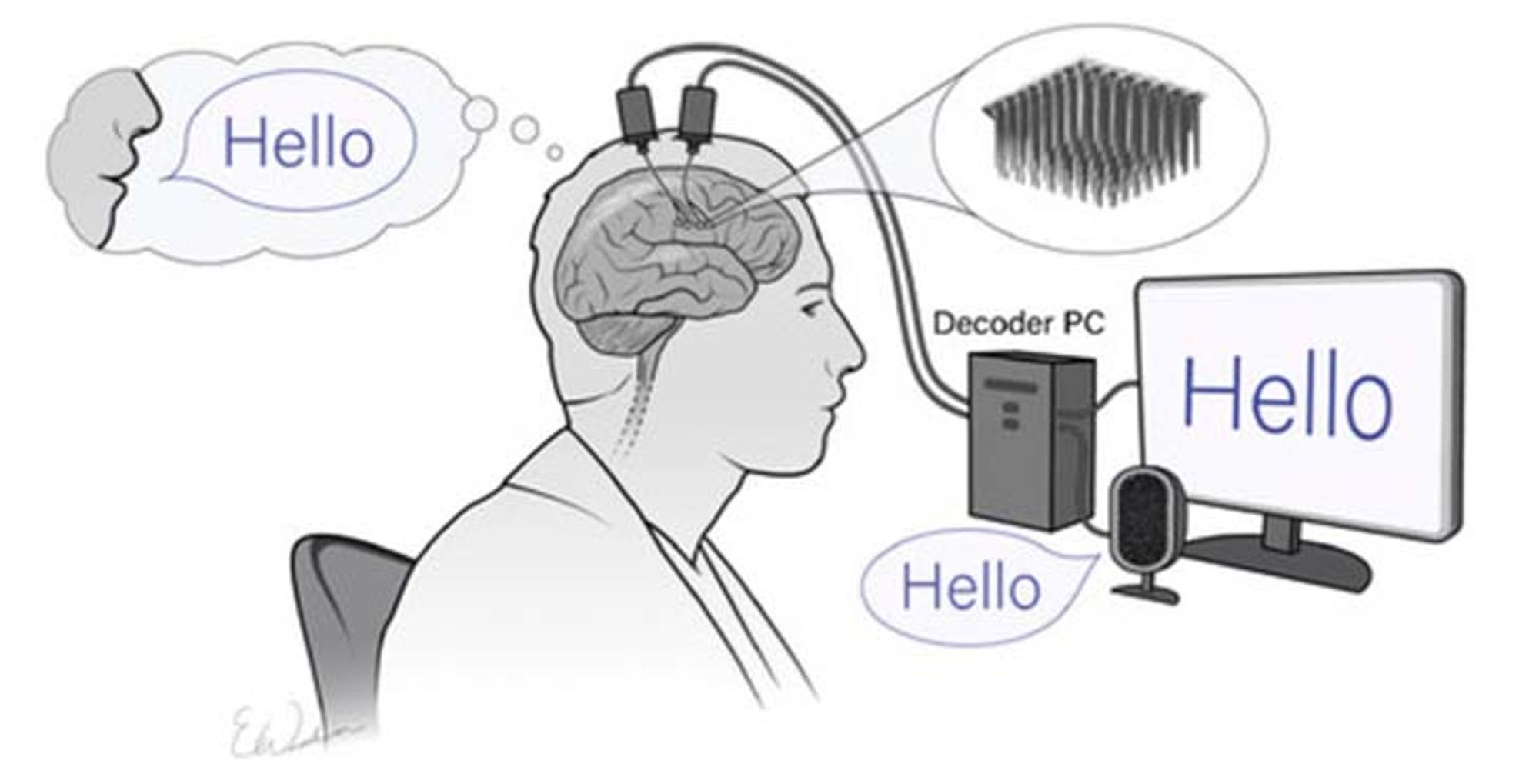
Sophisticated computer algorithms will be used to try to translate the patterns of brain cells involved in speaking into speech.
To turn the signals from nerve cells into the intended speech, the researchers will rely on advanced machine-learning techniques to create sophisticated computer algorithms. They anticipate these algorithms will be able to decode the neural patterns into speech accurately. The computer-decoded speech can then be paired with text-generating or synthetic speech-generating devices.
Stavisky notes that research groups are testing and improving these advanced medical devices that can potentially apply the recent progress in computing and neuroscience to provide new ways to treat paralysis.
“It is an exciting time. We still have much to learn, and there are no guarantees this will work. Based on how this field has been progressing and the strength of our interdisciplinary research team, I am hopeful that we can quickly make substantial progress towards restoring the ability for people who have lost the ability to speak to have naturalistic, real-time conversations,” Stavisky said.
Leigh Hochberg, director of the BrainGate consortium and clinical trial, said, “I’m thrilled to welcome David, Sergey and the wonderful neuroengineering community at UC Davis to the BrainGate clinical trials. Their scientific and clinical insights will undoubtedly lead to breakthroughs toward the restoration of communication.” Hochberg is a neurointensivist at Massachusetts General Hospital, a faculty member at Brown University and Harvard Medical School, and director of the VA RR&D Center for Neurorestoration and Neurotechnology at Providence VA Health System.
To be eligible, participants should:
- be over 18 and live within three hours of the greater Sacramento area.
- have paralysis in the arms and the legs or have difficulty speaking.
- have a medical diagnosis such as spinal cord injury, brainstem stroke, ALS or other degenerative motor neuron disorder.
To learn more about the study, visit Braingate.org or contact braingate@ucdavis.edu.
CAUTION: Investigational Device. Limited by Federal Law to Investigational Use.