Artificial polymer-based neural network. The strongly nonlinear behavior of these networks enables their use in reservoir computing. Credit: TU Dresden

Artificial intelligence (AI) will fundamentally change medicine and healthcare: Diagnostic patient data, e.g. from ECG, EEG or X-ray images, can be analyzed with the help of machine learning, so that diseases can be detected at a very early stage based on subtle changes. However, implanting AI within the human body is still a major technical challenge. TU Dresden scientists at the Chair of Optoelectronics have now succeeded for the first time in developing a bio-compatible implantable AI platform that classifies in real time healthy and pathological patterns in biological signals such as heartbeats. It detects pathological changes even without medical supervision. The research results have now been published in the journal Science Advances.
In this work, the research team led by Prof. Karl Leo, Dr. Hans Kleemann and Matteo Cucchi demonstrates an approach for real-time classification of healthy and diseased bio-signals based on a biocompatible AI chip. They used polymer-based fiber networks that structurally resemble the human brain and enable the neuromorphic AI principle of reservoir computing. The random arrangement of polymer fibers forms a so-called “recurrent network,” which allows it to process data, analogous to the human brain. The nonlinearity of these networks enables to amplify even the smallest signal changes, which – in the case of the heartbeat, for example – are often difficult for doctors to evaluate. However, the nonlinear transformation using the polymer network makes this possible without any problems.
In trials, the AI was able to differentiate between healthy heartbeats from three common arrhythmias with an 88% accuracy rate. In the process, the polymer network consumed less energy than a pacemaker. The potential applications for implantable AI systems are manifold: For example, they could be used to monitor cardiac arrhythmias or complications after surgery and report them to both doctors and patients via smartphone, allowing for swift medical assistance.
“The vision of combining modern electronics with biology has come a long way in recent years with the development of so-called organic mixed conductors,” explains Matteo Cucchi, PhD student and first author of the paper. “So far, however, successes have been limited to simple electronic components such as individual synapses or sensors. Solving complex tasks has not been possible so far. In our research, we have now taken a crucial step toward realizing this vision. By harnessing the power of neuromorphic computing, such as reservoir computing used here, we have succeeded in not only solving complex classification tasks in real time but we will also potentially be able to do this within the human body. This approach will make it possible to develop further intelligent systems in the future that can help save human lives.”
Reference: “Reservoir computing with biocompatible organic electrochemical networks for brain-inspired biosignal classification” by Matteo Cucchi, Christopher Gruener, Lautaro Petrauskas, Peter Steiner, Hsin Tseng, Axel Fischer, Bogdan Penkovsky, Christian Matthus, Peter Birkholz, Hans Kleemann and Karl Leo, 18 August 2021, Science Advances.DOI: 10.1126/sciadv.abh0693

Up to three cups of coffee per day is associated with a lower risk of stroke and fatal heart disease, according to research presented at ESC Congress 2021.1,2
“To our knowledge, this is the largest study to systematically assess the cardiovascular effects of regular coffee consumption in a population without diagnosed heart disease,” said study author Dr. Judit Simon, of the Heart and Vascular Centre, Semmelweis University, Budapest, Hungary.
“Our results suggest that regular coffee consumption is safe, as even high daily intake was not associated with adverse cardiovascular outcomes and all-cause mortality after a follow-up of 10 to 15 years,” she continued. “Moreover, 0.5 to 3 cups of coffee per day was independently associated with lower risks of stroke, death from cardiovascular disease, and death from any cause.”
Even though coffee is among the most consumed beverages in the world, little is known about the long-term impact of regular consumption on cardiovascular health.
This study investigated the association between usual coffee intake and incident heart attack, stroke, and death. The study included 468,629 participants of the UK Biobank with no signs of heart disease at the time of recruitment. The average age was 56.2 years and 55.8% were women.
Participants were divided into three groups according to their usual coffee intake: none (did not consume coffee on a regular basis, 22.1%), light-to-moderate (0.5 to 3 cups/day, 58.4%), and high (more than 3 cups/day, 19.5%).
The researchers estimated the association of daily coffee consumption with incident outcomes over a median follow-up of 11 years using multivariable models. The analyses were adjusted for factors that could influence the relationship including age, sex, weight, height, smoking status, physical activity, high blood pressure, diabetes, cholesterol level, socioeconomic status, and usual intake of alcohol, meat, tea, fruit and vegetables.
Compared to non-coffee drinkers, light-to-moderate consumption was associated with a 12% lower risk of all-cause death (hazard ratio [HR]=0.88, p<0.001), 17% lower risk of death from cardiovascular disease (HR=0.83, p=0.006), and 21% lower risk of incident stroke (HR=0.79; p=0.037).
To examine the potential underlying mechanisms, the researchers analyzed the association between daily coffee intake and heart structure and function over a median follow-up of 11 years. For this, they used data from 30,650 participants who underwent cardiac magnetic resonance imaging (MRI), which is considered the gold standard for the assessment of cardiac structure and function.
Dr. Simon said: “The imaging analysis indicated that compared with participants who did not drink coffee regularly, daily consumers had healthier sized and better functioning hearts. This was consistent with reversing the detrimental effects of aging on the heart.”
She concluded: “Our findings suggest that coffee consumption of up to 3 cups per day is associated with favorable cardiovascular outcomes. While further studies are needed to explain the underlying mechanisms, the observed benefits might be partly explained by positive alterations in cardiac structure and function.”
References and notes
- Abstract title: Association of daily coffee consumption with cardiovascular health – Results from the UK Biobank.
- Press conference: “Heart health made easy” on Thursday, August 26, from 17:00 to 18:00 CEST.
Funding: P.B.M and S.E.P acknowledge support from the National Institute for Health Research (NIHR) Barts Biomedical Research Centre. S.E.P. acknowledges support from the ‘SmartHeart’ EPSRC program grant (www.nihr.ac.uk; EP/P001009/1) and also from the CAP-AI program, London’s first AI enabling program focused on stimulating growth in the capital’s AI Sector. CAP-AI is led by Capital Enterprise in partnership with Barts Health NHS Trust and Digital Catapult and is funded by the European Regional Development Fund and Barts Charity. S.E.P. and S.N. acknowledge the British Heart Foundation for funding the manual analysis to create a cardiovascular magnetic resonance imaging reference standard for the UK Biobank imaging resource in 5000 CMR scans (www.bhf.org.uk; PG/14/89/31194). S.N and SKP supported by the Oxford NIHR Biomedical Research Centre and S.N. by the Oxford British Heart Foundation Centre of Research Excellence. N.A. recognizes the National Institute for Health Research (NIHR) Integrated Academic Training program which supports their Academic Clinical Lectureship posts. N.C.H acknowledges support from the UK Medical Research Council (MRC #405050259 and #U105960371), NIHR Southampton Biomedical Research Centre, University of Southampton, and University Hospital Southampton. Z.R.E was supported by a British Heart Foundation Clinical Research Training Fellowship (FS/17/81/33318). Project no. NVKP_16-1–2016-0017 (’National Heart Program’) has been implemented with the support provided from the National Research, Development and Innovation Fund of Hungary, financed under the NVKP_16 funding scheme. The research was financed by the Thematic Excellence Programme (2020-4.1.1.-TKP2020) of the Ministry for Innovation and Technology in Hungary, within the framework of the Therapeutic Development and Bioimaging thematic programs of the Semmelweis University.
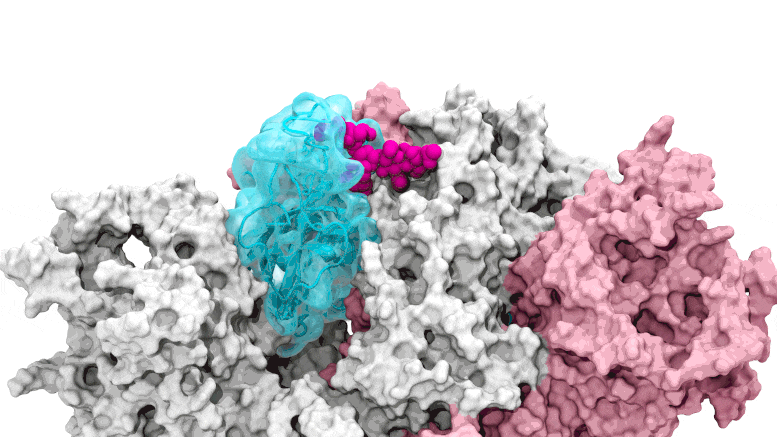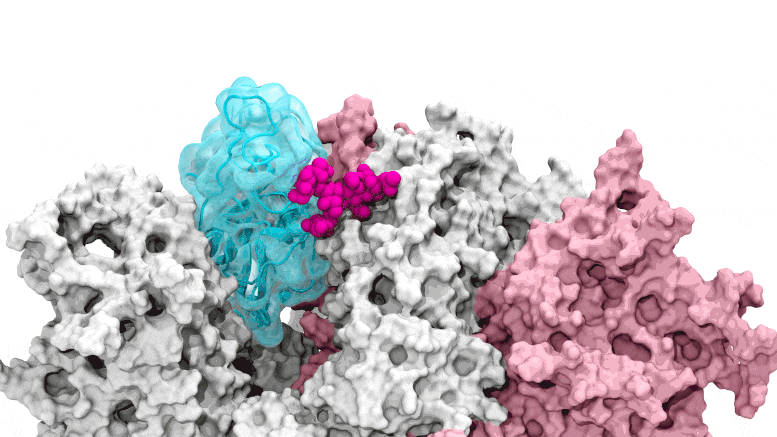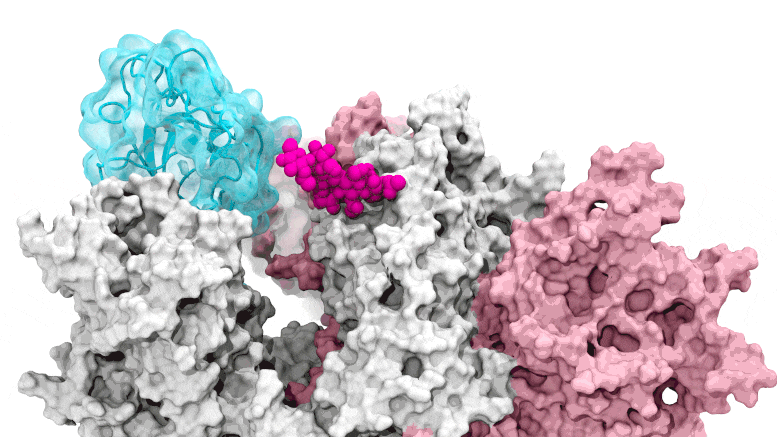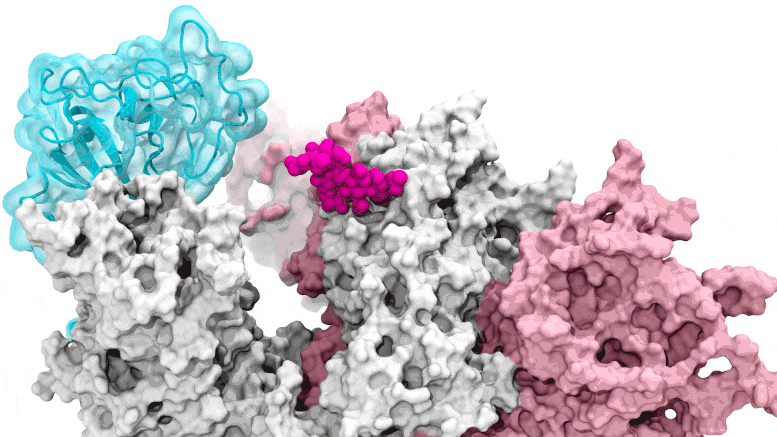
The glycan gate opens: Supercomputing-driven simulations depict the glycan N343 (magenta) acting as a molecular crowbar to pry open the SARS-CoV-2 spike’s receptor binding domain, or RBD (cyan), from a “down” to an “up” position. Credit: Terra Sztain, Surl-Hee Ahn, Lorenzo Casalino (Amaro Lab, UC San Diego)
Supercomputing-derived movies reveal details of deceptive sugar coating on spike protein, presenting new possibilities to block cell entry and infection.
Since the early days of the COVID pandemic, scientists have aggressively pursued the secrets of the mechanisms that allow severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to enter and infect healthy human cells.
Early in the pandemic, University of California San Diego’s Rommie Amaro, a computational biophysical chemist, helped develop a detailed visualization of the SARS-CoV-2 spike protein that efficiently latches onto our cell receptors.
Now, Amaro and her research colleagues from UC San Diego, University of Pittsburgh, University of Texas at Austin, Columbia University and University of Wisconsin-Milwaukee have discovered how glycans—molecules that make up a sugary residue around the edges of the spike protein—act as infection gateways.
Published August 19 in the journal Nature Chemistry, a research study led by Amaro, co-senior author Lillian Chong at the University of Pittsburgh, first author and UC San Diego graduate student Terra Sztain and co-first author and UC San Diego postdoctoral scholar Surl-Hee Ahn, describes the discovery of glycan “gates” that open to allow SARS-CoV-2 entry.
“We essentially figured out how the spike actually opens and infects,” said Amaro, a professor of chemistry and biochemistry and a senior author of the new study. “We’ve unlocked an important secret of the spike in how it infects cells. Without this gate the virus basically is rendered incapable of infection.”
Supercomputing-driven simulations depict the glycan N343 (magenta) acting as a molecular crowbar to pry open the SARS-CoV-2 spike’s receptor binding domain, or RBD (cyan), from a “down” to an “up” position. Credit: Terra Sztain, Surl-Hee Ahn, Lorenzo Casalino (Amaro Lab, UC San Diego)
Amaro believes the research team’s gate discovery opens potential avenues for new therapeutics to counter SARS-CoV-2 infection. If glycan gates could be pharmacologically locked in the closed position, then the virus is effectively prevented from opening to entry and infection.
The spike’s coating of glycans helps deceive the human immune system since it comes across as nothing more than a sugary residue. Previous technologies that imaged these structures depicted glycans in static open or closed positions, which initially didn’t draw much interest from scientists. Supercomputing simulations then allowed the researchers to develop dynamic movies that revealed glycan gates activating from one position to another, offering an unprecedented piece of the infection story.
“We were actually able to watch the opening and closing,” said Amaro. “That’s one of the really cool things these simulations give you—the ability to see really detailed movies. When you watch them you realize you’re seeing something that we otherwise would have ignored. You look at just the closed structure, and then you look at the open structure, and it doesn’t look like anything special. It’s only because we captured the movie of the whole process that you actually see it doing its thing.”
“Standard techniques would have required years to simulate this opening process, but with my lab’s ‘weighted ensemble’ advanced simulation tools, we were able to capture the process in only 45 days,” said Chong.
The computationally intensive simulations were first run on Comet at the San Diego Supercomputer Center at UC San Diego and later on Longhorn at the Texas Advanced Computing Center at UT Austin. Such computing power provided the researchers with atomic-level views of the spike protein receptor binding domain, or RBD, from more than 300 perspectives. The investigations revealed glycan “N343” as the linchpin that pries the RBD from the “down” to “up” position to allow access to the host cell’s ACE2 receptor. The researchers describe N343 glycan activation as similar to a “molecular crowbar” mechanism.
Jason McLellan, an associate professor of molecular biosciences at UT Austin and his team created variants of the spike protein and tested to see how a lack of the glycan gate affected the RBD’s ability to open.
“We showed that without this gate, the RBD of the spike protein can’t take the conformation it needs to infect cells,” McLellan said.
Reference: “A glycan gate controls opening of the SARS-CoV-2 spike protein” by Terra Sztain, Surl-Hee Ahn, Anthony T. Bogetti, Lorenzo Casalino, Jory A. Goldsmith, Evan Seitz, Ryan S. McCool, Fiona L. Kearns, Francisco Acosta-Reyes, Suvrajit Maji, Ghoncheh Mashayekhi, J. Andrew McCammon, Abbas Ourmazd, Joachim Frank, Jason S. McLellan, Lillian T. Chong and Rommie E. Amaro, 19 August 2021, Nature Chemistry.DOI: 10.1038/s41557-021-00758-3
The full author list includes: Terra Sztain, Surl-Hee Ahn, Anthony Bogetti, Lorenzo Casalino, Jory Goldsmith, Evan Seitz, Ryan McCool, Fiona Kearns, Francisco Acosta-Reyes, Suvrajit Maji, Ghoncheh Mashayekhi, J. Andrew McCammon, Abbas Ourmazd, Joachim Frank, Jason McLellan, Lillian Chong and Rommie Amaro.